Master 2 Internship Offer (6 months)
Exocube – Stability and preservation of cell envelopes under UV-exposure on Earth and in space
Key words: Exobiology, extremophiles (halophiles), biochemistry (lipids, proteins), spectroscopy/spectrometry/microscopy, Exocube (ISS experiment)
Supervisor: Dr Adrienne Kish
Lab: UMR 7245 MCAM (Molecules of Communication and Adaptation of Microorganisms), Muséum national d’Histoire naturelle, 63 rue Buffon, 75005 Paris
The preservation of microbial biosignatures in extreme conditions is crucial to distinguish traces of primitive life from abiotic organic signatures (on Earth or even on other planets). Microbial cell envelopes (cell wall + lipid membrane) have a strong potential to preserve biosignatures even over geological time scales (1). Environments considered extreme for human life, such as hypersaline environments, can challenge the stability of key microbial biomolecules such as the proteins and lipids of the cell envelope, but salts can also help to protect and preserve microbial cells and their structures. Evidence for past and present high salinity conditions are found throughout the solar system, from Earth to the icy moons of Enceladus and Europa (2, 3), to Mars and in meteorites (4). Evaporite crystals are known to be UV-shielding, particularly NaCl mixed with other salts such as KCl and MgSO4) (5). Carbon-based molecules of a non-biological origin have been identified preserved within the brine inclusions of extraterrestrial halite (NaCl) crystals found in meteorites (6). On Earth, evaporites such as halite are known to preserve organic molecules and even microorganisms. Living halophilic archaea have been isolated from the fluid inclusions of terrestrial Permo-Triassic halite (NaCl) crystals (7). Halophilic archaea such as Halobacterium salinarum are highly resistant to desiccation, high vacuum, UV-C, and gamma irradiation (8, 9) due to specific molecular adaptations that protect and repair damages to proteins and DNA. While halophilic archaea have shown varying degrees of resistance to simulated and actual space conditions (10–12), little data is currently available concerning the molecular-level effects of solar radiation exposure on their key biomolecules, such as cell envelopes. The cell envelope of halophilic archaea is a simple structure but resistant to extreme conditions: a cell membrane composed of ether-linked lipids with isoprenoid chains surrounded by a proteinaceous surface layer (S-layer) of self-assembled into a symmetrical 2-D array.
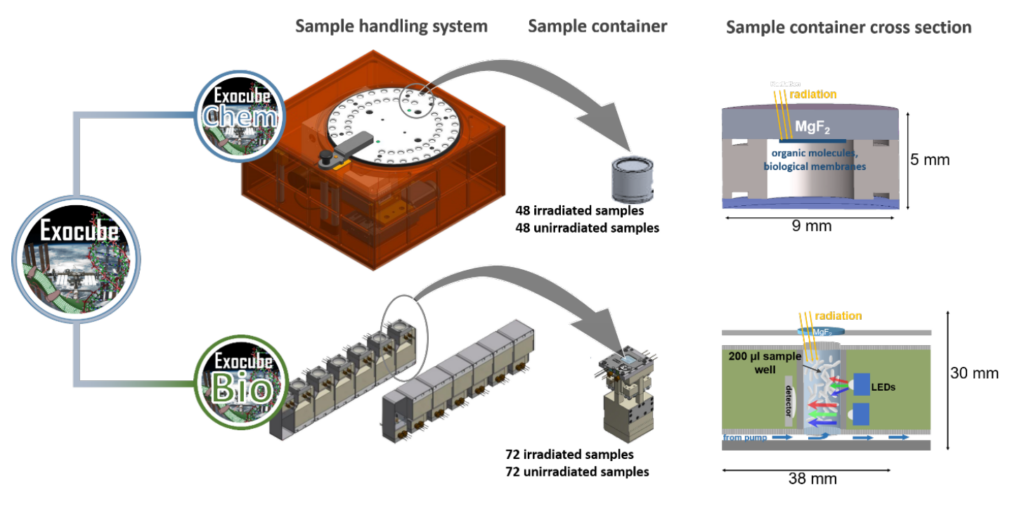
The goal of this Master 2 project is to evaluate the structural stability of the cell envelopes of halophilic archaea under high-UV conditions, up to and including simulations of UV-conditions in Low Earth Oribt (LEO) in preparation for the launch of the Exocube experiment. The Exocube space exposure platform (see Fig. 1) is currently being developed by the European Space Agency as part of a new European Space Exposure Platform (EXPO) for the International Space Station(13). The Exocube project aims to gain insights into the biological and photochemical stability of life’s building blocks and cell surface components. Results from Exocube will help to interpret findings from past and upcoming planetary exploration and lifedetection missions in our solar system and possibly beyond. The intern will compare the stability of cell envelope extracts and those of whole cells (living and dead) both within and outside of halite crystals. Samples will be exposed to full UV irradiation (200-400nm) using solar simulator equipped with a high-pressure xenon-arc lamp. Protocols for in-flight analyses (fluorescence/FT-IR spectroscopies) and post-flight analyses (atomic force/electron microscopies) of cell envelope integrity will be tested, in collaboration with the Exocube principal investigator, Dr Andreas Elsaesser at the Freie Universität Berlin.
Candidates should have a background in microbiology and or biochemistry, and a strong interest in exobiology.
Contact: Dr Adrienne Kish
References:
1. A. Kish et al., Scientific reports. 6, 26152–10 (2016).
2. S. K. Trumbo, M. E. Brown, K. P. Hand, Science advances. 5, eaaw7123 (2019).
3. F. Postberg et al., Nature. 474, 620–622 (2011).
4. S. W. Squyres et al., Science. 313, 1403–1407 (2006).
5. P. F. W. Morwool, thesis (2019).
6. Q. H. S. Chan et al., 4, eaao3521 (2018).
7. H. Stan-Lotter et al., Int J Astrobiol. 1, 271–284 (2002).
8. M. Kottemann, A. Kish, C. Iloanusi, S. Bjork, J. DiRuggiero, Extremophiles. 9, 219–227 (2005).9. A. Kish et al., Environ Microbiol. 11, 1066–1078 (2009).
10. A. Oren, 372 (2014), doi:10.1098/rsta.2014.0194.
11. K. Olsson-Francis et al., Method Microbiol, 27–65 (2018).
12. S. Leuko, P. Rettberg, Astrobiology. 17, 110–117 (2017).
13. A. Sgambati et al., Acta Astronaut. 166, 377–390 (2019).




Aucun commentaire sur l'article Master 2 Internship Offer on the stability and preservation of cell envelopes under UV (Paris)